Chemical energy is a form of potential energy that will only be observed when it is released in a chemical reaction. Chemical energy is the energy of chemical bonds and is also stored in atoms and ions.
Interestingly, when chemical energy is released, the substance from which the energy came is often changed into an entirely new substance. Many everyday items store and release chemical energy. Let's take a look at examples of chemical energy to see just how often we interact with this scientific wonder.
9 Examples of Chemical Energy
Chemical energy is either released (exothermic reaction) or absorbed (endothermic reaction) during a chemical reaction. In an exothermic reaction, heat is released, creating warmth. In an endothermic reaction, the heat is absorbed, creating cooling.
- Air bags are activated by a chemical reaction inside the bag. A sensor turns on an electrical circuit, and then sodium azide is ignited. The reaction that occurs generates nitrogen gas, which fills the bag at an extremely rapid rate.
- Ammonia and bleach when mixed create a chemical reaction. Toxic vapors develop into a chemical known as chloramine vapor. This has the potential to form hydrazine which can cause edema, headaches, nausea, and seizures.
- Baking soda and vinegar produce carbon dioxide gas when mixed. As this gas grows in volume, it puts pressure on whatever container it is in, and will erupt out of an opening in the container, creating a volcano effect. This chemical reaction is safe, making it a great science activity for kids.
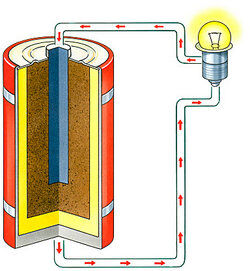
- Batteries have two terminals: the anode and the cathode. They're separated by an electrolyte, a chemical that allows the anode and cathode to transmit a charge. When something's connected to a battery, chemical reactions take place along the electrodes to produce electricity. Of course, you can't see the energy in the battery when the battery is just sitting around; it is when the electricity is produced that the energy is seen.
- Explosives: when an explosive goes off, chemical energy that was stored in it is changed and transferred into sound energy, kinetic energy, and thermal energy. Just because chemical energy is being released, it doesn't mean an explosion will occur. But, when a solid material quickly changes into a hot, expanding gas, you may be looking at an explosion. Take TNT, for example. Two units of solid TNT can instantly change into 15 units of hot gas, creating a dark and sooty explosion.
- Food: chemical energy in food is released when the food is digested and the molecules of food are broken down into smaller pieces. Our bodies digest food by mixing it with acids and enzymes in the stomach. This process turns carbohydrates into glucose. The stomach (and small intestine) then release that glucose into the bloodstream, serving as energy for our bodies.
- Heating packs (used to warm up cold hands or soothe sore muscles) have chemicals inside them. A lot of them function by "cracking" (or bending) them. This is because they're filled with iron. Once you crack the heating pack, the iron is exposed to the air. As the iron reacts to the oxygen, it transforms into iron oxide, a chemical that can produce heat.
- Petroleum is a combination of oil and natural gas made of hundreds of molecules containing carbon and hydrogen. When petroleum is a vapor, it's considered natural gas. When petroleum is a liquid, it's considered crude oil. We then extract that energy by burning it and using it to fuel cars, heat homes, and more.
- Wood, when dry, stores chemical energy. This chemical energy is released as the wood burns, and it is converted into heat, or thermal energy. This also produces light energy. As a result of burning, the wood turns into an entirely new substance - ashes.
Everyday Chemistry
Isn't it interesting to see how many types of chemical energy we interact with every day? The very wood we burn and the airbags we face every time we enter a vehicle are examples of chemical energy. This seems like a nice opportunity for a new hypothesis to form.
Looking for more scientific discovery? What do you know about radiant energy? It's energy that travels via waves or particles. These examples of radiant energy will introduce you to the concept. Let the love of science continue!