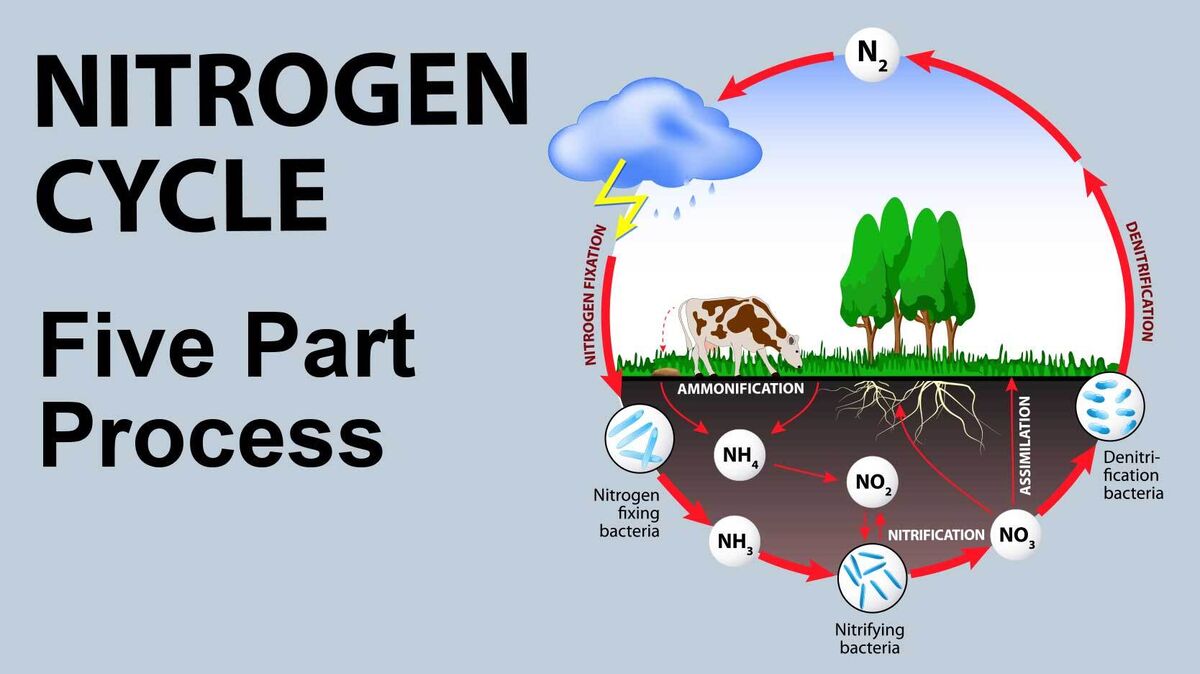
The term nitrogen cycle refers to the process through which nitrogen transforms into its various states or forms. It is a five-part process that can occur through biological or physical means. Discover the basics of the process and review some nitrogen cycle examples.
Understanding the Nitrogen Cycle Steps
Because so much of the Earth's atmosphere consists of nitrogen (greater than ¾), it is extremely important for scientists to understand the nitrogen cycle in order to determine and comprehend its effects on various ecosystems.
- nitrogen fixation - Bacteria that contain nitrogenase convert nitrogen (which is a gas) from the atmosphere so that it becomes ammonia.
- nitrification - Bacteria in the soil convert the ammonia to nitrogen dioxide. Another type of bacteria then adds an oxygen atom, which creates nitrate.
- assimilation - Plants convert nitrate to chemicals they need, such as nucleic and amino acids. Animals that eat the plants then utilize these substances.
- ammonification - When plants and animals die, the ammonification process causes the nucleic and amino acids to turn back into ammonia and nitrates. They are then released back into the soil.
- denitrification - Bacteria convert nitrates back into nitrogen, which is then released back into the atmosphere.
Real-World Nitrogen Cycle Examples
Some examples of the nitrogen cycle include the following.
- A plant takes nitrogen from the soil by absorbing it through its roots. The nitrogen comes in the form of nitrogen ions. When the nitrogen is absorbed by the plant, it is reduced to nitrite ions. Next, it becomes ammonium ions which can be incorporated into amino or nucleic acids and into chlorophyll.
- When a plant dies or an animal dies or when a plant or an animal expels waste, organic nitrogen is then released. Bacteria are able to convert this organic nitrogen into ammonium. They do this through a process called mineralization.
- Nitrogen gets into the oceans as a result of runoff from groundwater or when it rains. Nitrogen can also get into the ocean through precipitation (rain). Nitrogen in the water undergoes fixation, which is generally facilitated by a type of bacteria called cyanobacteria. After fixation, the nitrogen is in a biologically available form that phytoplankton in the ocean can use.
- Plankton excrete urea and ammonia into the water. Phytoplankton and waste products can sink downward, introducing ammonia at a depth below the euphotic zone. The ammonia from the waste products is then removed from the euphotic zone and bacteria that live below the euphotic zone can convert the ammonia into nitrate. Once the ammonia is converted, nitrification occurs and the ammonia becomes nitrite and nitrate. Vertical mixing and upwelling can carry the nitrate upward and it can then be used by phytoplankton to continue the cycle.
Facilitating the Nitrogen Cycle
The nitrogen cycle is affected by factors that work to facilitate the conversion of nitrogen into various states and back through the atmosphere. These factors include:
- the use of agricultural fertilizer
- the growing of legumes by agriculturalists.; legumes are able to convert nitrogen to nitrates and nitrites
- the use of nitrates by bacteria in soil
- microbes in soil and water that change ammonia into nitrites
- bacteria called Nitrosomonas that convert ammonia to nitrites
- bacteria called Nitrobacter change nitrites to nitrates
- termites and shipworms pair with bacteria to change nitrogen's form
- cyanobacteria that live in semi-aquatic environments can participate in the nitrogen cycle
- industrial fixation can be used to convert nitrogen to ammonia at temperatures of 600 degrees and beyond with the use of catalysts
- nitrogen can be fixed atmospherically via lightning
A Life-Sustaining Process
Now you have lots of different examples of how the nitrogen cycle works and of some of the things that can impact the nitrogen cycle. These examples should help you better understand this important and life-sustaining process. To expand your scientific knowledge, next explore some examples of natural succession of plant life. From there, consider studying some examples of organic compounds.